Thyroid optimization — DTE-first
Desiccated thyroid extract as a first-line option, the FT3-driven optimal-target framing, the levothyroxine-to-DTE conversion algorithm, and the documentation rule that protects against the FDA Boxed Warning.
Learning objectives
After completing this module, you will be able to:
- Differentiate the Nimbus FT3-driven optimal-target framing from a TSH-driven, T4-monotherapy model and explain the deiodinase rationale.
- Apply the DTE initiation protocol (0.5–1 grain QAM, 0.25 grain in AFib/SVT, monthly titration, max 2 grains BID).
- Construct the levothyroxine → DTE conversion (100 mcg ≈ 1 grain DTE; double the DTE dose at 2 weeks to compensate for T4 storage depletion).
- Interpret why exogenous TSH suppression in a patient with euthyroid Free T3 and resolved symptoms is not iatrogenic Graves' disease.
- Evaluate when to add liothyronine (T3) instead of escalating DTE above 4 grains/day, and how to dose it.
- Apply the documentation rule — never record weight loss as a thyroid indication — that protects against the FDA Boxed Warning on all thyroid hormones.
Disclosures & accreditation statement
Activity type
Internal Provider Education
Faculty & planners
- Dr. Jobby John, PharmD, FACA (author) — Founder & CEO, Nimbus Healthcare (employer); Inventor of IntelliHealth Clinical Decision Intelligence Platform.
- Dr. Jobby John, PharmD, FACA (reviewer) — Founder & CEO, Nimbus Healthcare (employer); primary clinical reviewer.
- Dr. Richard Harris, MD, PharmD, MBA (reviewer) — Secondary clinical reviewer. Relevant financial relationships: [to be disclosed].
Off-label / unapproved use
This activity discusses off-label or unapproved use of: Desiccated thyroid extract (Armour, NP Thyroid, WP Thyroid, compounded) for hypothyroidism — grandfathered, not formally FDA-evaluated for safety/efficacy, Liothyronine (synthetic T3) as combination add-on therapy with DTE or levothyroxine, Thyroid replacement dosed to optimal Free T3 (not to TSH) in symptomatic patients whose TSH/FT4 fall within standard reference ranges, Compounded T3-only and T4+T3 combinations beyond labeled indications, Thyroid replacement for prevention endpoints (CV risk, cognitive decline, dyslipidemia, metabolic syndrome) outside of biochemical hypothyroidism.
Commercial support
None.
All relevant financial relationships have been identified, reviewed, and mitigated per ACCME Standards for Integrity and Independence.
The premise
Thyroid hormone is the body's pace-setter. Every cell expresses thyroid receptors; the hormone tunes basal metabolic rate, body temperature, cardiac output, lipid handling, mood, cognition, and reproductive function. The active hormone at the receptor is Free T3, not Free T4 and not TSH. T4 is largely a pro-hormone — it has to be converted to T3 by a family of selenium-dependent enzymes called 5′-deiodinases before it does meaningful work at the cellular level.
There are three deiodinase types, and you need to know what each one does because the distinction is what justifies treating the way Nimbus treats:
- Type I (D1): liver, kidney, thyroid. Generates circulating T3 and also drives reverse-T3 inactivation. Sensitive to propylthiouracil.
- Type II (D2): CNS, pituitary, retina, heart, skin, skeletal muscle, brown adipose. The dominant route by which peripheral tissues make their own intracellular T3 from T4. High circulating T4 down-regulates D2, which means T4-monotherapy at high doses can paradoxically lower active tissue T3.[bianco-2019]
- Type III (D3): CNS, skin, placenta. Inactivates T3 to reverse T3. Up-regulated by illness, stress, starvation, certain drugs.
Disruptors of deiodinase activity that come up in clinical practice: propylthiouracil, beta-blockers, high-iodide loads, endocrine disruptors and heavy metals, starvation, hypothyroidism itself, uncontrolled diabetes, uremia. The practical implication is that serum T4 (even Free T4) does not reliably predict the T3 the tissues actually see, and TSH — set by the pituitary's local D2 — does not reliably predict tissue T3 either.
This module trains the prescriber to (1) target Free T3 and symptoms rather than TSH, (2) prefer desiccated thyroid extract (DTE) over levothyroxine monotherapy, (3) run the levothyroxine-to-DTE conversion correctly, and (4) follow a documentation rule that protects the practice from the FDA Boxed Warning every thyroid hormone label carries.
Pre-test — anchor on the target
Check yourself
The Nimbus optimal Free T3 range for an adult on thyroid therapy is approximately:
Thyroid physiology — TSH, FT4, FT3, the deiodinase types
Image
Thyroid anatomy. The 1.5–2 cm² gland produces T4 (mostly) and T3 (minority); peripheral deiodinase generates the remaining ~80% of circulating T3.
Credit·National Cancer Institute (NCI) / SEER Cancer Training Modules, public domain (US Government work under 17 U.S.C. § 105)
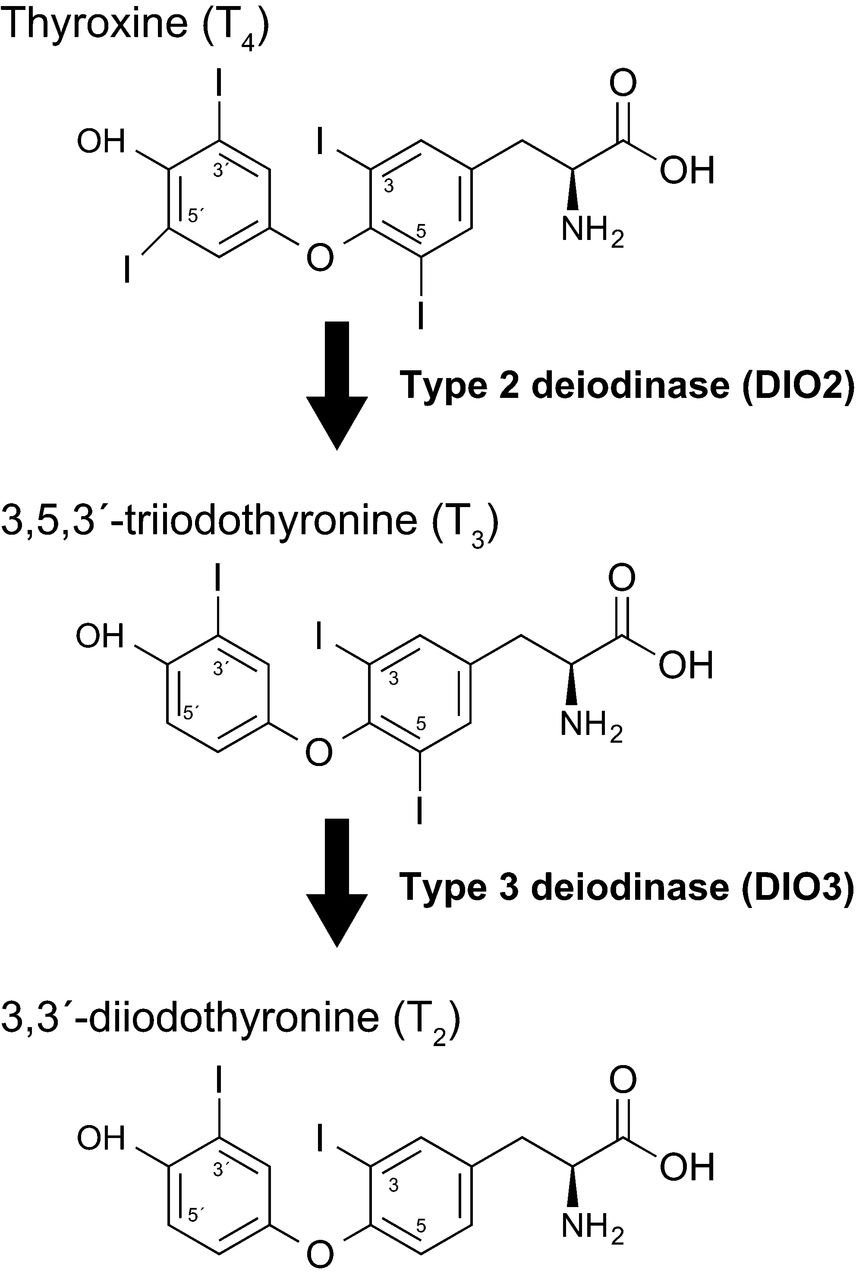
Image
Deiodinase types D1, D2, and D3 control peripheral T4→T3 conversion (D1 + D2 activate; D3 inactivates). High T4 (typical on levothyroxine monotherapy) suppresses D2 in many peripheral tissues — the mechanism underlying why some patients remain symptomatic at 'optimal' TSH on T4-only.
Credit·Taeko Nishiwaki-Ohkawa and Takashi Yoshimura, CC BY-SA 4.0 (share-alike) — https://creativecommons.org/licenses/by-sa/4.0/
The classic hypothalamic-pituitary-thyroid (HPT) axis: TRH → TSH → thyroid gland → T4 (~80%) and T3 (~20%). Most T3 the tissues use is generated peripherally from T4 by D1 and D2. The pituitary's TSH set-point reflects its own local D2 activity — which is why a "normal" TSH does not guarantee adequate T3 elsewhere in the body.
Two clinical consequences of the deiodinase architecture matter on every visit:
- T4-only therapy is biochemically incomplete. High Free T4 down-regulates D2; over months, peripheral T3 falls even as the lab "looks good." This is why some levothyroxine patients remain symptomatic with a TSH inside the standard reference range.[hoang-2013]
- T3-only therapy suppresses TSH and ultimately starves the system of T4 reserve. D2 needs substrate. Long-term T3 monotherapy produces a hyper-then-hypo pattern.
The Nimbus default is replace both — DTE if the patient has no pork allergy or religious restriction, otherwise levothyroxine + liothyronine.
Numeric targets
Every thyroid follow-up at Nimbus reads off the same target sheet. Memorize this table.
Analyte
TSH on therapy (mIU/L)
- Lab reference0.4–4.5 mIU/L
Nimbus optimal: 0.3–1 mIU/L
Figure
The Nimbus thyroid lab targets. FT3 drives the titration; TSH is the noisiest of the three (diurnal variation ~17% in women); FT4 may decline as FT3 is optimized and that is intended — not a failure.
Source·Nimbus clinical policy; BHRT syllabus; supporting literature in docs/anti-gap/05-thyroid.md
Analyte
Free T3 (pg/mL)
- Lab reference2.3–4.2 pg/mL5
Nimbus optimal: 4–4.3 pg/mL
Figure
Free T3 is the active hormone at the receptor. The Nimbus target sits in the upper third of the reference range; young adults may run up to 5.0 pg/mL.
Source·Nimbus clinical protocol module 05; reference ranges Quest / LabCorp 2026
Analyte
Free T4 (ng/dL)
- Lab reference0.8–1.8 ng/dL
Nimbus optimal: 0.8–1.4 ng/dL
Figure
Free T4 may decline as FT3 is optimized on DTE — this is INTENDED, not a failure of titration. Within reference is sufficient; do not drive FT4 up at the expense of FT3 quality.
Source·Nimbus clinical protocol module 05; reference ranges Quest / LabCorp 2026
TSH (on therapy)
0.3–1.0mIU/L
Mainstream lab range 0.3–5.0; classic AACE proposal 0.3–2.0. We target the lower third. TSH alone is the least reliable single indicator.
Free T3
4.0–4.3pg/mL
Up to ~5.0 pg/mL in young adults. The active hormone — drive the dose to this plus symptoms.
Free T4
Within reference
May decline as FT3 optimizes on DTE — not the gating metric.
LT4 → DTE equivalence
100 mcg≈ 1 grain DTE
Anchor for the conversion algorithm; double the DTE dose at the 2-week mark.
DTE start
0.5–1 grainQAM
0.25 grain in AFib/SVT. Empty stomach, 30 min before food/drink/meds.
DTE ceiling
2 grains BID
If still under-replaced at 4 grains/day total, add liothyronine 5–25 mcg — do NOT keep escalating DTE.
DTE-first rationale — why combination T4+T3 outperforms T4 monotherapy
DTE (Armour, NP Thyroid, WP Thyroid, compounded equivalents) is desiccated porcine thyroid. It contains T4 and T3 in the natural porcine ratio plus small amounts of T1 and T2. Patients absorb and respond to it differently than to synthetic T4 alone or to synthetic T4 + T3 split tablets — most patients feel better, sleep better, and report clearer cognition on DTE than on equivalent T4 monotherapy.[hoang-2013-pref]
The randomized literature on T4+T3 combination therapy versus T4 monotherapy is mixed in the aggregate but consistent in one place: in the symptomatic subgroup who did not feel well on levothyroxine alone, combination therapy improved well-being and quality-of-life measures.[bunevicius-1999] The Wiersinga synthesis of the European Thyroid Association working group reaches the same practical conclusion: a structured trial of combination therapy is reasonable in the persistently symptomatic patient.[wiersinga-2012]
Initiation — start low, titrate by symptoms
The DTE titration ladder is the load-bearing piece of this module. The timeline below shows the cadence at a glance; the widget underneath walks through it interactively; the prose covers the AFib carve-out, the twice-daily dosing decision, and the rule that stops escalation at 4 grains/day.
Figure
The Nimbus DTE titration ladder. Monthly cadence; FT3 + symptoms drive each step; AFib/SVT carve-out at initiation; T3 add-on at the cap rather than DTE escalation.
Source·Nimbus clinical protocol; BHRT syllabus; conversion equivalence 100 mcg levothyroxine ≈ 1 grain DTE
Interactive · DTE titration ladder
Walk a hypothetical patient up the ladder
Daily total
1.5 gr
~ 150 mcg LT4 equiv
Schedule
1.5 grain QAM
Clinical guardrail
Re-test TSH + Free T3 at 4–6 weeks before further escalation.
Standard adult start: 0.5–1 grain DTE PO QAM, empty stomach, 30 minutes before food, drink, or other medications. Plain water is fine; if local water is high-mineral, distilled is preferable for the morning dose window.
Cardiac carve-out: AFib history, SVT history, or known cardiac sensitivity → start 0.25 grain QAM and titrate by 0.25 grain monthly rather than the standard 0.5–1 grain monthly. Slower is safer; the goal is symptom improvement without provoking arrhythmia.
Up-titration: by 0.5–1 grain monthly (0.25 grain monthly in cardiac patients) until Free T3 is in the 4.0–4.3 pg/mL band and symptoms resolve. Re-check TSH + Free T3 + Free T4 at 4–6 weeks after each dose change.
Twice-daily dosing: if afternoon fatigue persists at 1.5–2 grains QAM, split the dose (AM + early-PM). Cap the second dose at 2–3 PM — later than that and DTE drives insomnia.
Ceiling and the T3 add-on rule: the maximum is 2 grains BID (4 grains/day). If symptoms persist at 4 grains/day, add liothyronine 5–25 mcg rather than continuing to escalate DTE — at higher DTE doses, additional T4 stops adding peripheral T3 because of D2 down-regulation.
Levothyroxine → DTE conversion algorithm
This is the most frequently mis-run conversion in DTC. The trap: a clinician converts at "equivalent" dose and the patient feels worse at week 2 because the stored T4 pool that was buffering them has run out.
The algorithm:
- Confirm the patient is truly hypothyroid (has been on levothyroxine for an evidenced indication) or is a symptomatic euthyroid-by-labs patient electing the off-label trial — document accordingly.
- Stop levothyroxine.
- Start DTE at the equivalent dose: 100 mcg levothyroxine ≈ 1 grain DTE. So 50 mcg → 0.5 grain; 100 mcg → 1 grain; 150 mcg → 1.5 grain.
- At 2 weeks: double the DTE dose. This compensates for T4 storage depletion. Without this step the patient crashes between weeks 2 and 6.
- Continue titrating by symptoms and Free T3 every 4–6 weeks until at target. Twice-daily dosing if afternoon fatigue persists.
Mid-module checkpoint
Check yourself
A patient on 100 mcg levothyroxine daily wants to switch to DTE. What is the correct conversion?
Never use SR formulations of thyroid hormone
Sustained-release (SR) thyroid preparations impair absorption and produce inconsistent serum levels through the day. Nimbus does not use SR thyroid in any form (DTE, levothyroxine, or liothyronine). The same rule applies to progesterone. SR is reserved for melatonin and DHEA where the pharmacokinetic profile actually serves the indication.
TPO antibody testing — optional, not required
Many mainstream protocols routinely order TPO antibodies. The Nimbus position: TPO testing is optional. A positive TPO confirms Hashimoto's, but it does not change the treatment plan when symptoms plus Free T3 are already driving the dose. Order it when the differential matters for counselling (e.g., a young patient considering pregnancy) or when the patient asks; otherwise it is not part of the baseline panel.
Treatment-induced TSH suppression is NOT iatrogenic Graves' disease
This is the rule the model and the prescriber both need to internalize: a patient who is on titrated DTE with euthyroid Free T3, no palpitations, no tremor, no weight loss, no insomnia, and resolved symptoms is not in iatrogenic hyperthyroidism even if the TSH is suppressed. The classic risks attributed to TSH suppression in the literature — atrial fibrillation, bone loss, sudden cardiac death — derive from autoimmune thyrotoxicosis (Graves' disease) where Free T3 and Free T4 are both elevated and the patient is symptomatic. They do not transfer cleanly to symptom-optimal exogenous DTE dosing in which Free T3 is in range.
Documentation discipline — never document weight loss as the indication
Every thyroid hormone in the US carries the FDA Boxed Warning that thyroid hormones are not indicated for the treatment of obesity or weight loss and that larger doses may produce serious or life-threatening toxicity, particularly when given with sympathomimetic amines.[fda-thyroid-bbw]
The documentation rule is therefore absolute: never record weight loss as the indication for thyroid replacement. If a patient mentions weight loss as the reason for asking, redirect: thyroid replacement is for symptomatic hypothyroid function documented by labs and clinical findings; for weight management we route to the GLP-1 / WeightWise tier with appropriate documentation. If the same patient also has documented hypothyroid symptoms with a low Free T3, document that indication separately and treat it on its own merits.
Role-specific operations
Preview · showing both tracks
For prescribers
Initiation workflow. Baseline labs: TSH, Free T4, Free T3 (plus the panel from module 00). Decide the indication — documented hypothyroidism vs. symptomatic euthyroid-by-labs trial — and document it on initiation. Confirm no AFib/SVT (use 0.25 grain start if present), no untreated adrenal insufficiency (replace cortisol first), and no active hyperthyroidism. Start 0.5–1 grain DTE QAM, empty stomach, 30 minutes before anything else. Re-check TSH + Free T3 + Free T4 at 4–6 weeks.
Titration cadence. Up-titrate by 0.5–1 grain monthly (0.25 grain in cardiac patients) until Free T3 is 4.0–4.3 pg/mL and symptoms resolve. Split to AM + early-PM if afternoon fatigue persists. Cap the second dose at 2–3 PM.
Levo → DTE conversion. Stop levothyroxine. Start DTE at the equivalent dose (100 mcg ≈ 1 grain). At 2 weeks, double the DTE dose to compensate for T4 storage depletion. Titrate to symptoms and Free T3 thereafter.
T3 add-on decision. If symptoms persist at 4 grains DTE/day, add liothyronine 5–25 mcg rather than continuing to escalate DTE. Split the liothyronine across AM + early-PM if half-life behaviour produces afternoon dips. Re-check labs at 4–6 weeks.
FT3 monitoring cadence. Every 4–6 weeks after each titration; every 3 months on a stable dose for the first year; annually thereafter unless symptoms change. Always include Free T3 — TSH alone is not sufficient.
For pharmacists
DTE brand options. Armour Thyroid (Allergan), NP Thyroid (Acella), WP Thyroid (RLC Labs — historical supply interruptions; check current availability), and compounded DTE for patients who need a specific dose increment or excipient profile. Compounded DTE allows non-grain doses (e.g., 0.75 grain) and excipient customization for patients with lactose, gluten, or dye sensitivities.
Bioequivalence considerations. The grain → mcg-equivalent labelling differs subtly between manufacturers because of how T4 + T3 content is assayed and reported. Counsel patients that switching brands at the same nominal grain dose can shift perceived effect; if you must switch (supply issue), recheck labs at 4–6 weeks.
AM empty-stomach counselling. Take on rising, with plain water, 30 minutes before food, drink (including coffee), other medications, and supplements. Distilled water if local water is high-mineral. Calcium, iron, magnesium, and PPI acid-blockers reduce thyroid absorption — separate by at least 4 hours.
Drug-interaction profile. Calcium and iron (separate by 4 hours), PPIs (reduce absorption), aluminum-containing antacids (separate by 4 hours), bile acid sequestrants (separate by 4 hours), estrogen (increases TBG and may raise total T4 — but Free T3 / Free T4 typically remain stable), warfarin (thyroid increases sensitivity — monitor INR after dose changes).
Never SR. Reject any prescription for sustained-release thyroid. Same rule for sustained-release progesterone. SR is reserved for melatonin and DHEA.
Cheat sheet — the numbers that drive the visit
Optimal TSH on therapy
0.3–1.0mIU/L
Treat-to-FT3, not to TSH; do not reduce dose on a low TSH alone.
Optimal Free T3
4.0–4.3pg/mL
Up to ~5.0 pg/mL in young adults.
Standard DTE start
0.5–1 grainQAM
Empty stomach, 30 min before food/drink/meds.
Cardiac carve-out
0.25 grainQAM
AFib, SVT, cardiac sensitivity — titrate 0.25 grain monthly.
DTE ceiling
2 grains BID
Then add liothyronine 5–25 mcg — do not escalate DTE further.
LT4 ↔ DTE
100 mcg ≈ 1 grain
Doubling rule at 2 weeks to compensate for T4 storage depletion.
Conversion doubling
× 2 at 2 weeks
Without it the patient crashes between weeks 2 and 6.
Formulation rule
Never SR
No sustained-release thyroid in any form.
Documentation rule
Never weight loss
FDA Boxed Warning on every thyroid hormone label.
TSH suppression
≠ Graves'
With euthyroid FT3 and resolved symptoms, suppressed TSH on therapy is not iatrogenic hyperthyroidism.
Post-test — synthesis
Check yourself
A 48-year-old woman has been on 100 mcg levothyroxine daily for 4 years. She reports persistent fatigue, cold intolerance, and brain fog. Labs: TSH 1.2 mIU/L, Free T4 1.3 ng/dL (within range), Free T3 2.8 pg/mL (low end of reference). She has no AFib history. What is the best next step?