Testosterone optimization in men
Subcutaneous TRT with the saturation-model framing, % free PSA gating, the under-40 fertility-first protocol, and the cases where AI / 5α-RI co-prescription is wrong.
Learning objectives
After completing this module, you will be able to:
- Differentiate the saturation-model framing of androgen action from a linear dose-response model and apply it when reasoning about borderline-PSA TRT decisions.
- Apply the subcutaneous TRT initiation protocol (80–100 mg/wk default, +10–20 mg q4–6 wk titration, cap 160 mg/wk) and select alternate routes when SC is not appropriate.
- Interpret PSA workup using percent-free PSA cutoffs (<10% high suspicion, 10–20% indeterminate, >20% unlikely) and the 3-day pre-draw prep rules.
- Construct an under-40 hypogonadism workup that prioritizes fertility (clomiphene or hCG before TRT) and rules out pituitary adenoma (prolactin >40 ng/mL).
- Evaluate when aromatase inhibitor and 5α-reductase inhibitor co-prescription with TRT harms more than it helps, and articulate the dose/frequency-first alternative.
- Apply the hematocrit gating protocol (≥52% reduce dose, ≥54% HOLD, phlebotomy and OSA workup as needed) and the topical transference protocol.
Disclosures & accreditation statement
Activity type
Internal Provider Education
Faculty & planners
- Dr. Jobby John, PharmD, FACA (author) — Founder & CEO, Nimbus Healthcare (employer); Inventor of IntelliHealth Clinical Decision Intelligence Platform.
- Dr. Jobby John, PharmD, FACA (reviewer) — Founder & CEO, Nimbus Healthcare (employer); primary clinical reviewer.
- Dr. Richard Harris, MD, PharmD, MBA (reviewer) — Secondary clinical reviewer. Relevant financial relationships: [to be disclosed].
Off-label / unapproved use
This activity discusses off-label or unapproved use of: Subcutaneous testosterone cypionate / enanthate (FDA label is intramuscular), Compounded testosterone cream applied to scrotum (commercial gel labels prohibit genital application), Compounded testosterone pellets up to 1,600 mg per insertion cycle (commercial Testopel is 75 mg/pellet), Clomiphene 25–50 mg QOD for male hypogonadism with intact fertility, hCG 500–1,500 IU SC 2–3×/week for endogenous T preservation in men with intact fertility, Anastrozole low-dose for symptomatic estrogen excess refractory to TRT dose/frequency adjustment (on-label indication is breast cancer), TMP-SMX 800/160 mg PO q12h × 14 days for empiric prostatitis when PSA elevation is suspected inflammatory.
Commercial support
None.
All relevant financial relationships have been identified, reviewed, and mitigated per ACCME Standards for Integrity and Independence.
The premise
Testosterone replacement therapy is the cleanest "optimization" intervention in the Nimbus catalogue — and the easiest to get wrong by reflex. The pre-2024 era was dominated by fear of cardiovascular and prostate-cancer harm. TRAVERSE 2023[TRAVERSE-2023] randomized 5,204 men with hypogonadism and preexisting or high CV risk to transdermal testosterone vs. placebo and found no increase in major adverse cardiovascular events. The FDA removed the MACE black-box warning in February 2025 (with a new caution that some oral testosterone products may raise blood pressure).
What's left is a gating problem. The Nimbus protocol gets the gating right by anchoring on three ideas that mainstream urology often does not foreground:
- The saturation model — prostate androgen receptors saturate at roughly 250 ng/dL total testosterone. Therapeutic TRT above that does not proportionally increase prostate-cancer risk, so "his testosterone is high" is not by itself a reason to hold.
- Free T over Total T — total T is bound to SHBG and albumin and does not always correlate with symptoms. Free T does. A 48-year-old with Total T 926 ng/dL and low libido can still be hypogonadal at the free-T level.
- Fertility first under 40 — men under 40 with hypogonadism get a clomiphene or hCG trial before exogenous T, plus a prolactin to rule out pituitary adenoma.
Pre-test — anchor the saturation model
Check yourself
Per the Nimbus saturation-model framing, above approximately what total testosterone level is the prostate androgen receptor saturated, such that additional testosterone does not further stimulate prostate growth?
TRT physiology — the saturation model, total vs. free, why E2 still matters
Schematic
Marks et al. demonstrated prostate androgen receptors saturate at approximately 250 ng/dL. Above the saturation point, additional testosterone does not increase prostate stimulus — the basis for the Nimbus position that hypogonadal men should be treated to physiologic Free T regardless of total-T anxiety.
Source·Conceptual rendering of the Marks LS et al. saturation-model framework (JAMA 2006, PMID 17105798) and subsequent reanalyses including Morgentaler / Traish / Khera reviews.
Testosterone circulates ~98% bound (SHBG, albumin) and ~2% free. The free fraction is what drives androgen-receptor signaling and symptom resolution. Total T is misleading when SHBG is high (aging, hyperthyroidism, oral E2) or low (insulin resistance, obesity). The Nimbus protocol gates on Free T, not Total T.
Inside the prostate, the androgen receptor saturates at approximately 250 ng/dL total testosterone. Past that point additional T does not further accelerate prostate growth — which is the rational basis for not reflexively holding TRT on the strength of a high T level alone.
A second physiologic anchor: testosterone aromatizes to estradiol. In men, E2 is the dominant regulator of bone health — androgen-deprivation states show bone, brain, and CV consequences that track E2 suppression more than T suppression[Falahati-Nini-2000]. That is why routine aromatase inhibition is not a Nimbus default (see the AI section below).
Initiation — subcutaneous is the default
Subcutaneous testosterone is the Nimbus first-line. Patients self-administer in the abdomen or thigh with an insulin-style syringe. Compared to intramuscular: smaller needle, less peak-trough swing, easier home use, better adherence. The FDA label for testosterone cypionate / enanthate is IM — subcutaneous use is off-label, with the rationale and counseling documented per Nimbus policy.
SC starting dose
80–100mg/wk
Once weekly default; some patients split into 2× weekly to flatten peaks.
Titration cadence
+10–20mg q4–6 wk
Adjust to symptoms + Free T + Hct, not chasing Total T alone.
Cap
~160mg/wk
Above this, side-effect burden (Hct, edema, E2) typically outweighs incremental benefit.
Daily SC option
~30mg/day
Alternative when weekly produces symptomatic peak-trough; smoothest E2.
Saturation anchor
~250ng/dL total T
Above this, prostate AR is saturated — additional T does not further stimulate prostate growth.
Free T target (Quest)
170–210pg/mL
Lab 'normal' 50–210 is not optimal; treat toward the upper end with symptoms.
Free T target (LabCorp)
30–40pg/mL
Different assay; use vendor-aware targets, not a single number.
Hct HOLD
≥54%
≥52 reduce dose; ≥54 HOLD pending OSA workup ± phlebotomy.
Reassess at 8–12 weeks with symptoms, Free T, Hct, and PSA (if ≥40). Symptom resolution within the optimal Free T range is the endpoint — do not chase a single lab number.
Alternate routes
| Route | Typical dose | Notes |
|---|---|---|
| Subcutaneous T cypionate / enanthate | 80–100 mg/wk; or ~30 mg/day | Nimbus default. Off-label vs. IM label. |
| Intramuscular T cypionate / enanthate | 100–200 mg IM q1–2 wk | On-label. Peak-trough swings; HDL may drop year 1. |
| Compounded T cream to scrotum | <200 lb: 100 mg/gm 0.5–1 gm BID; >200 lb: 200 mg/gm 0.5–1 gm BID | Fastest absorption, highest DHT, no alcohol sting. Off-label site. Apply ~4 hr before bedtime to minimize transference. |
| Commercial transdermal gel (Androgel 1% / 1.62%, Fortesta, Vogelxo) | 10–100 mg/day; shoulders/upper arms/abdomen/thighs | On-label. Alcohol base — flammable until dry. Transference risk to partners/children/pets. Avoid genitals/chest/back per label. |
| Oral testosterone undecanoate — Kyzatrex / Jatenzo / Tlando | Kyzatrex 100–200 mg BID (max 400 mg BID); Jatenzo 158/198/237 mg BID; Tlando 112.5 mg × 2 caps BID | Lymphatic absorption; BID with food. FDA label cautions BP elevation on oral T. |
| Long-acting T undecanoate (Aveed) | 750 mg IM, second dose at 4 wk, then q10 wk | REMS-restricted; in-office post-injection monitoring for POME (pulmonary oil microembolism). |
| Pellets (Testopel + compounded) | 600–1,200 mg typical; 1,400–1,600 mg in larger / chronic-disease men; inserted q3–6 mo | High compliance, no transference. Off-label pellet doses. |
| Patch (Testoderm, Androderm) | — | Discontinued commercially. |
| Buccal (Striant) | — | Discontinued — gum irritation, taste perversion. |
| Axillary solution (Axiron) | — | Rarely used. |
Free T target — Quest vs. LabCorp
The single most common mistake in TRT interpretation is reading a Free T value without asking which vendor produced it. Quest and LabCorp use different free-T assay methodologies and report different "normal" ranges. The Nimbus optimal targets are vendor-aware: roughly 170–210 pg/mL on Quest, 30–40 pg/mL on LabCorp. Use the converter below as a quick check before dose adjustments.
Interactive · Quest ↔ LabCorp converter
Progesterone (postmeno)
Equivalent on quest
10.50 ng/mL
Optimal target · Quest >= 10 · LabCorp >= 3
Bioidentical progesterone. Do not test in pre/peri/PCOS — treat by symptoms.
PSA gating — percent-free PSA and the prostatitis pathway
Pre-TRT baseline PSA + digital rectal exam (DRE) in men ≥40 with risk factors or ≥55 generally. Annual PSA on TRT at minimum. 3-day pre-draw prep: no vigorous exercise, no cycling, no sexual activity, no DRE — all of which transiently raise PSA.
If PSA exceeds 2.5 ng/mL on TRT, recheck and order percent-free PSA. The decision tree below mirrors the Nimbus protocol:
Interactive · Percent-free PSA decision
Where does this patient land on the protocol?
Repeat & consider referral
Percent-free PSA 10–20% is indeterminate. Repeat PSA + percent-free in 3–4 months; consider urology consultation if the trend persists. Confirm 3-day prep (no vigorous exercise, cycling, sex, DRE) before re-draw.
Suspected prostatitis as the cause of a PSA bump gets an empiric course of TMP-SMX 800/160 mg PO q12h × 14 days (extend to 21–30 days if persistent); alt cephalexin 500 mg PO q12h × 14 days. Retest PSA 3–4 months after completion. Caveats on PSA interpretation:
- 5α-reductase inhibitors (finasteride / dutasteride) lower PSA roughly 50% with no effect on percent-free.
- Statins, NSAIDs, acetaminophen, and thiazides also pull PSA down.
- Recent sex, exercise, cycling, DRE, instrumentation, or a hard bowel movement transiently push PSA up.
Urology referral triggers: PSA rise >1.4 ng/mL above baseline within 12 months; PSA
>4.0 ng/mL; abnormal DRE; percent-free PSA <10%.
Check yourself
A 58-year-old man on TRT for 18 months has a baseline PSA of 1.0 ng/mL. At today's annual draw his PSA is 3.2 ng/mL. He confirms he completed a 100-mile cycling event two days ago. Per the Nimbus PSA pathway, what is the best next step?
Hct gating — and the OSA workup hidden inside it
The most common dose-limiting side effect of TRT is hematocrit rise. The Nimbus protocol:
- Hct ≥52% — reduce dose or extend interval; consider blood donation.
- Hct ≥54% — HOLD therapy; phlebotomy if symptomatic; workup OSA (STOP-Bang).
- Untreated severe OSA is the most common reversible cause; high-altitude residence (>~3,000 m / 10,000 ft) is a benign physiologic adaptation and may not require intervention.
Figure
Hct gating on TRT. The HOLD threshold is non-negotiable — secondary erythrocytosis on testosterone is the most common reversible cause of TRT cessation. STOP-Bang ≥3 mandates sleep evaluation; OSA is a common occult driver.
Source·Nimbus clinical protocol §Hct gating; framing aligned with Bhasin S et al. Endocrine Society guideline 2018 (PMID 29562364) and Coviello AD et al. for TRT-erythrocytosis epidemiology.
Frame this for the patient as a workup gate, not a punishment — the secondary erythrocytosis is usually telling you something about sleep-disordered breathing.
Under-40 men — fertility first, prolactin to rule out adenoma
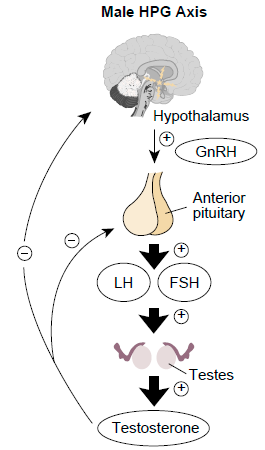
Image
The hypothalamus → pituitary → testis axis. GnRH drives LH pulses; LH stimulates Leydig-cell testosterone; T provides negative feedback at both upstream points. Exogenous TRT suppresses the entire axis — the basis for the Nimbus under-40 fertility-first protocol.
Credit·Kong L, Zhang T, Tang M, Wang D, CC BY 4.0 — derivative diagram from Int. J. Mol. Sci. 2014;15(11):21253–21269 (DOI 10.3390/ijms151121253), reproduced via Wikimedia Commons. https://creativecommons.org/licenses/by/4.0/
Mainstream practice often treats hypogonadism the same way regardless of age. Nimbus diverges: men under 40 with confirmed symptomatic hypogonadism get a clomiphene or hCG trial before exogenous testosterone. Reasoning:
- Exogenous T suppresses GnRH → LH/FSH → spermatogenesis; testicular atrophy and infertility follow.
- Clomiphene 25–50 mg QOD (off-label) is a SERM that increases endogenous LH/FSH and preserves spermatogenesis.
- hCG 500–1,500 IU SC 2–3×/week (off-label) directly stimulates Leydig cells.
- Both are reversible. Exogenous T's fertility effects are not always reversible.
Concurrent with the fertility decision, check prolactin. Prolactin >40 ng/mL in a young hypogonadal man is a red flag for pituitary adenoma — work that up before any androgen therapy.
Man under 40 with hypogonadal symptoms + low T
Confirm symptoms + 2× AM total T below threshold
Check prolactin
Pituitary rule-out before any androgen therapy
STOP — pituitary workup
MRI sella, endocrinology referral; defer androgen therapy until cleared
Fertility intact + actively trying to conceive?
Document the fertility conversation either way
Clomiphene OR hCG (NOT TRT)
Clomiphene 25–50 mg QOD OR hCG 500–1,500 IU SC 2–3×/wk — preserves spermatogenesis
Discuss fertility risk; clomiphene/hCG first-line
TRT acceptable only if patient prefers it AND consents to fertility tradeoff
Schematic
Under-40 men with hypogonadal symptoms get the prolactin rule-out and the fertility conversation BEFORE any TRT decision. Clomiphene and hCG preserve endogenous LH/Leydig-cell function; TRT suppresses both.
Source·Nimbus clinical protocol — under-40 male hypogonadism workup; clomiphene-for-male-hypogonadism literature including Hsieh TC et al. 2013 (PMID 23260550) and the IntelliHealth V6 BHRT syllabus framing.
AI in men — why routine use is wrong
Routine aromatase inhibitor co-prescription with TRT is one of the most common "optimization" errors. Estradiol in men is not a side effect to be suppressed — it is the dominant regulator of bone, is required for libido / cognition / joint comfort, and its suppression in androgen-deprivation patients drives the worst quality-of-life sequelae[Finkelstein-2013].
The Nimbus approach when E2 is symptomatic:
- Reduce dose — many "high E2" symptoms resolve at a lower T dose.
- Increase frequency — splitting weekly into 2× weekly (or going daily SC) flattens the E2 peak.
- Reserve low-dose AI (e.g., anastrozole) only for symptomatic estrogen excess refractory to (1) and (2), and time-limit it (1–2 months, then reassess).
Counsel patients about the costs of routine AI: low libido, ED, mood lability, visceral fat gain, bone loss, joint pain, increased CAD/CVA/mortality signals when used routinely in men.
5α-RI in men — finasteride / dutasteride are not first-line on TRT
Routine co-prescription of finasteride or dutasteride with TRT to prevent hair loss is not a Nimbus default. Two reasons:
- DHT is required for orgasm and sexual function. Blunting it can erase a meaningful fraction of the TRT benefit.
- Post-finasteride syndrome (PFS) — persistent sexual, neurological, and physical adverse effects after discontinuation, potentially irreversible. Counseling about PFS is required before any 5α-RI Rx.
If a patient explicitly wants a 5α-RI for hair loss after counseling and optimization of ferritin (>80) and thyroid: finasteride 1 mg/day (lowers PSA ~50% with no effect on percent-free) or dutasteride 0.5 mg twice weekly. Topical minoxidil 5% (or oral minoxidil 1.25 mg/day) is the first-line hair-loss option.
TRAVERSE — what the cardiovascular safety data actually showed
TRAVERSE 2023[TRAVERSE-2023] randomized 5,204 hypogonadal men aged 45–80 with preexisting or high CV risk to 1.62% transdermal testosterone vs. placebo, mean follow-up 21.7 months. The primary endpoint (composite of CV death, non-fatal MI, non-fatal stroke) was non-inferior in the T arm. There was a small numeric increase in atrial fibrillation, acute kidney injury, and pulmonary embolism — flagged in labeling but not enough to support the pre-2025 MACE black-box.
The clinical implication is conservative but real: in symptomatic hypogonadal men with or without CV risk, TRT does not measurably worsen MACE within roughly 2 years of treatment. The FDA removed the MACE black-box in February 2025 and added a BP-caution, particularly for newer oral testosterone products.
TODO(citation): AUA TRT guideline (Mulhall et al. 2018, updated) for the mainstream comparator framing of Total T as primary gating diagnostic. Search PubMed when finalizing.
TODO(citation): Tomic et al. for clomiphene in male hypogonadism — confirm PMID before publication.
TODO(citation): Yassin et al. 2023 long-term TTh in hypogonadal men (LUTS endpoint) — confirm PMID.
TODO(citation): McHenry et al. 2014 review on testosterone, mood, and depression — confirm PMID.
TODO(citation): Jung 2016 prospective placebo-controlled trial of TRT for cognition in hypogonadal men — confirm PMID.
TODO(citation): Okada 2016 IM testosterone enanthate / LUTS — confirm PMID.
Transference — the household-contact counseling
Topical testosterone transfers to partners, children, and pets via skin contact and contaminated fabrics. Transference is teratogenic to a fetus. Counsel every topical patient:
- Apply ~4 hours before bedtime (the Nimbus default for scrotal cream).
- Wash hands thoroughly with soap after application.
- Use separate towels and washcloths from children and pregnant / nursing partners.
- Wash clothes and bedsheets separately.
- Not an issue with injectables, orals, or pellets — only topicals.
If a household partner is pregnant or nursing, switch route (SC injection is the simplest swap) or apply with the strictest transference protocol.
Track branches
Preview · showing both tracks
For prescribers
Initiation and titration. Default SC cypionate 80–100 mg/wk after the workup
gates clear (Free T below optimal + symptoms; PSA + DRE if ≥40; STOP-Bang; prolactin
if <40; baseline Hct, CMP, lipids). Titrate +10–20 mg q4–6 wk to symptom resolution
within the optimal Free T window; cap at ~160 mg/wk.
PSA workflow. Annual PSA on TRT. 3-day prep before every draw. PSA >2.5 → recheck + percent-free PSA. Use the decision tree above for the referral call. Suspected prostatitis → TMP-SMX 800/160 q12h × 14 d, retest at 3–4 months.
Hct gating. ≥52% reduce dose; ≥54% HOLD + OSA workup + phlebotomy if needed.
Under-40 protocol. Clomiphene 25–50 mg QOD or hCG 500–1,500 IU SC 2–3×/wk before exogenous T. Prolactin to rule out pituitary adenoma. Document fertility counseling on every initial TRT Rx regardless of age.
Avoid by default. Routine AI co-prescription. Routine 5α-RI co-prescription. Reflexive TRT holds on the basis of "high T" without the PSA / percent-free pathway.
For pharmacists
Compounding — scrotal cream. Two strength tiers: 100 mg/gm (under 200 lb) and 200 mg/gm (over 200 lb), dosed 0.5–1 gm BID. Verify base compatibility (some bases sting). Counsel: apply ~4 hours before bedtime to minimize transference. Document off-label site rationale on the script.
SC injection technique counseling. Insulin syringe in abdomen or thigh, rotate sites, no aspiration needed at SC depth. Once weekly default; split into 2× weekly if peak-trough symptoms. Daily SC ~30 mg/day for the smoothest E2 profile.
Transference counseling on every topical fill. Separate towels, separate laundry, wash hands. Teratogenic to a fetus — if a household partner is pregnant or nursing, escalate to the prescriber for a route change.
Dose conversion across routes. Patients who switch from IM weekly to SC weekly keep the mg/wk; patients who switch to daily SC divide by 7. Patients who switch to a commercial gel need explicit re-counseling on application sites (not genitals / chest / back) and on alcohol-base flammability while wet.
Oral T (Kyzatrex / Jatenzo / Tlando). BID with food required (lymphatic absorption). FDA label warns oral T may raise BP — flag baseline / on-therapy BP monitoring back to the prescriber.
Pellet considerations. Compounded pellets exceed Testopel 75 mg/pellet — common Nimbus dose 600–1,200 mg (larger / chronic-disease men 1,400–1,600 mg) inserted q3–6 months. Off-label pellet doses; document rationale.
Post-test — synthesis
Check yourself
A 52-year-old man on TRT 100 mg/wk SC for 14 months returns for annual labs. Free T 195 pg/mL (Quest), Hct 53%, PSA 1.9 ng/mL up from 1.2 ng/mL twelve months ago, BP 128/82, STOP-Bang 4 (untreated). He has no urinary symptoms. Best next step per the Nimbus protocol?